
10.3 forbindelser af chlor Kemi Libretekster Anne Marie
0:00 / 2:24 Lewis Structures, Introduction, Formal Charge, Molecular Geometry, Resonance, Polar or Nonpolar A step-by-step explanation of how to draw the ClO2 - Lewis Dot Structure.
Clo2 1 Lewis Structure
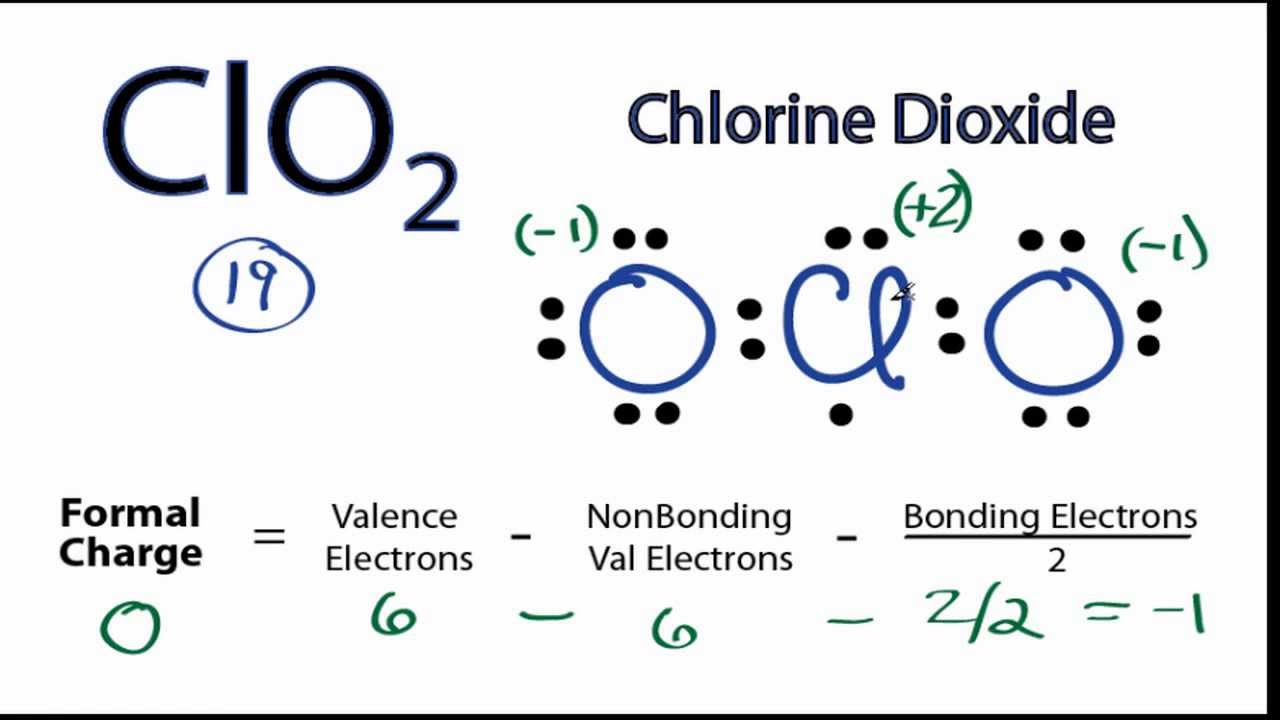
ClO 2- lewis structure has a Chlorine atom (Cl) at the center which is surrounded by two Oxygen atoms (O). There is 1 single bond and 1 double bond between the Chlorine atom (Cl) and each Oxygen atom (O). There are 2 lone pairs on double bonded Oxygen atom (O) and 3 lone pairs on single bonded Oxygen atom (O).

How can I draw the Lewis structure for ClO2?
Hey Guys,We are back with one of the most requested videos, ClO2 Lewis structure. Chlorine Dioxide is made up of one chlorine atom and two oxygen atoms. To u.

So far, we’ve used 8 of the Cl2 Lewis structure’s total 14 outermost valence shell electrons
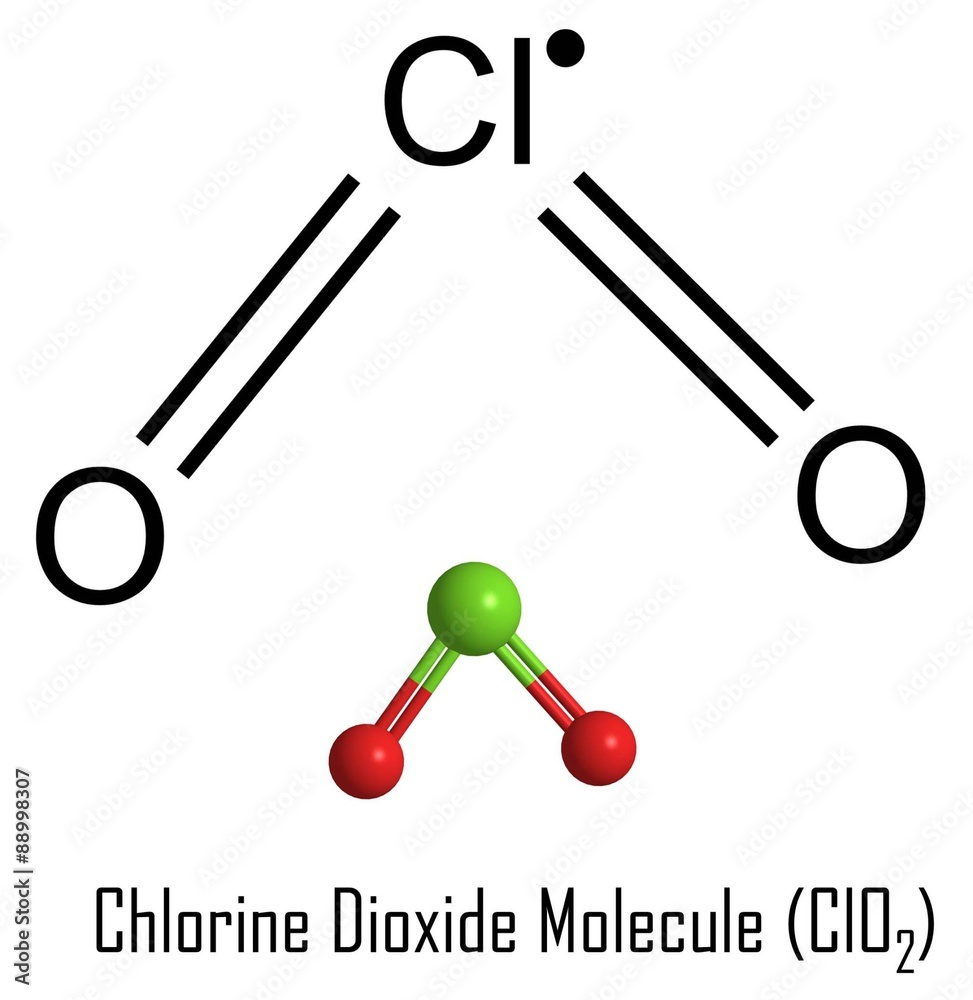
Lewis structure of ClO2 (or Chlorine dioxide) contains two double bonds between the Chlorine (Cl) atom and Oxygen (O) atom. The Chlorine atom (Cl) is at the center and it is surrounded by 2 Oxygen atoms (O). The Chlorine atom has 1 lone pair and 1 unpaired electron, while both the Oxygen atoms have 2 lone pairs..

What is the bond order of ClO2?
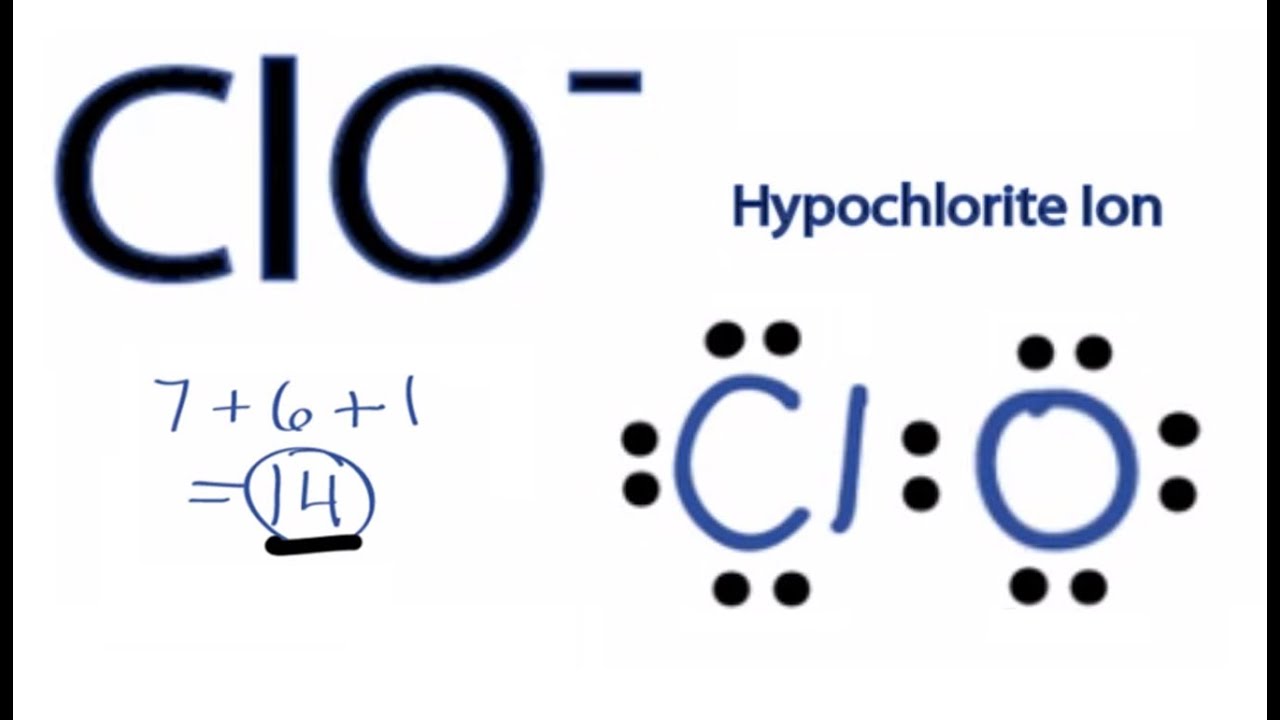
ClO2- lewis structure comprises two oxygen (O) atoms and one chlorine (Cl) atom. The chlorine (Cl) atom is kept at the central position and the oxygen (O) atoms are in the surrounding position in the lewis diagram. The lewis dot structure of ClO2- contains 7 lone pairs and 3 bonded pairs.

ClO2 Lewis Structure How to Draw the Lewis Structure for ClO2 (Chlorite Ion) YouTube
A quick explanation of the molecular geometry of ClO2 - (Chlorite ion) including a description of the ClO2 - bond angles.Looking at the ClO2- Lewis structure.

Strutture di Lewis • Ione Clorito [ClO2] YouTube
Lewis Structure of Chlorine Dioxide (ClO2-) The Lewis structure is a pictorial representation of valence electrons taking part in the formation of bonds to produce a new molecule with new properties altogether. To begin drawing the Lewis structure of Chlorine dioxide, first, it is essential to draw one for the participating elements. For Chlorine,

How do we draw Lewis dot structure for ClO2 with proper explanation please Chemistry
In the Lewis structure for ClO2- we put Chlorine (Cl) at the center of the structure since it is the least electronegative. There are total of 20 valence electrons for the ClO2- Lewis structure. Remember that the negative sign counts as one valence electron.
ClO2 Lewis Structure How to Draw the Lewis Structure for ClO2 YouTube
Lewis structure of ClO2- (or Chlorite ion) contains one double bond and one single bond between the Chlorine (Cl) atom and Oxygen (O) atom. The Chlorine atom (Cl) is at the center and it is surrounded by 2 Oxygen atoms (O). The Chlorine atom has 2 lone pairs, one Oxygen atom has 2 lone pairs and the other oxygen atom has 3 lone pairs.

Clo2 1 Lewis Structure
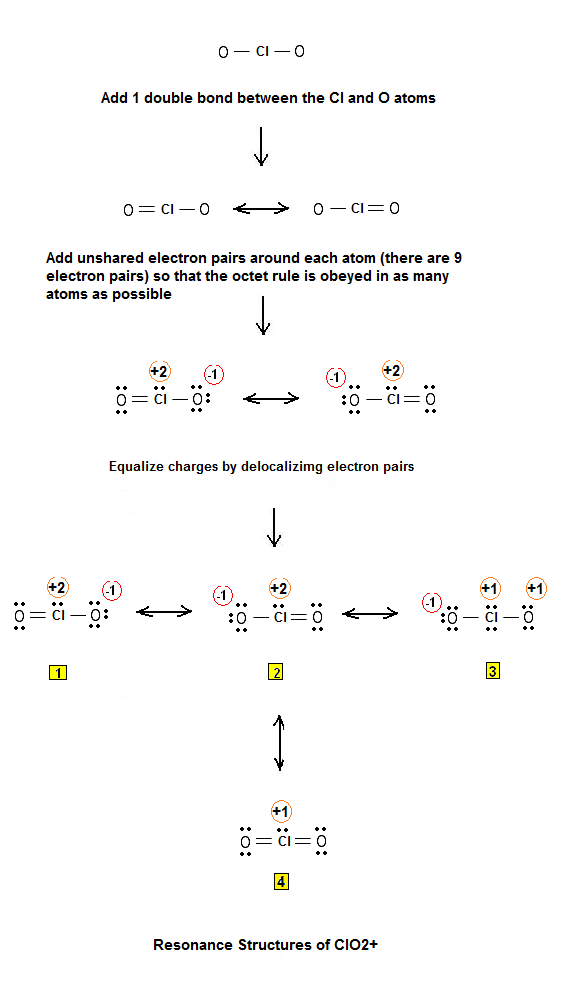
The trial structure is You have 20 valence electrons in your trial structure. The valence electrons you have available are: 1 Cl + 2 O + 1 e = 1 ×7 +2 ×6 + 1 = 20. Hence, the trial structure has the correct number of electrons. The formal charge on each atom is: Cl = 7 − 4 −½(4) = +1;O = 6- 6 −½(2) = -1 Every atom has a formal charge.
Clo2 1 Lewis Structure
In the Lewis structure of ClO 2, there are two double bonds around the chlorine atom, with two oxygen atoms attached to it. Each oxygen atom has two lone pairs, and the chlorine atom has one lone pair and one unpaired electron. How to Draw the Lewis Structure for ClO2 (Chlorine dioxide) Watch on Contents Steps #1 Draw a rough skeleton structure

Clo2 1 Lewis Structure
A step-by-step explanation of how to draw the ClO2 Lewis Dot Structure (Chlorine dioxide)The ClO2 Lewis structure has 19 valence electrons meaning that there.

How Can I Draw The Lewis Structure For Clo2 Class 11 Chemistry Cbse Images and Photos finder
ClO2 lewis structure has a Chlorine atom (Cl) at the center which is surrounded by two Oxygen atoms (O). There are 2 double bonds between the Chlorine atom (Cl) and each Oxygen atom (O). There are 2 lone pairs on both the Oxygen atoms (O) and 1 lone pair & 1 unpaired electron on the Chlorine atom (Cl).

ClO2 Lewis Structure, Geometry, Hybridization, and Polarity Techiescientist
Drawing the Lewis Structure for ClO 2. For the Lewis structure for ClO 2 place Chlorine (Cl) in the center of the structure since it is the least electronegative. ClO 2 has an odd number of valence electrons (19). That means that the Chlorine (Cl) atom will have an unpaired electron and therefore won't have an octet (it will have seven in ClO 2 .
Lewis ElectronDot Structure for Chlorine Dioxide Ion (ClO2+) Chemistry Net
In the ClO 2- Lewis structure, there is one single bond and one double bond around the chlorine atom, with two oxygen atoms attached to it. The oxygen atom with a single bond has three lone pairs, and the oxygen atom with a double bond has two lone pairs. Also, there is a negative (-1) charge on the oxygen atom with a single bond. Contents Steps

Clo2 1 Lewis Structure
I quickly take you through how to draw the Lewis Structure of ClO2- (Chlorite Ion). I also go over the formal charge, hybridization, shape, bond angle and re.