/nitrogenaton-58b602813df78cdcd83d8348.jpg)
Atoms Diagrams Electron Configurations of Elements
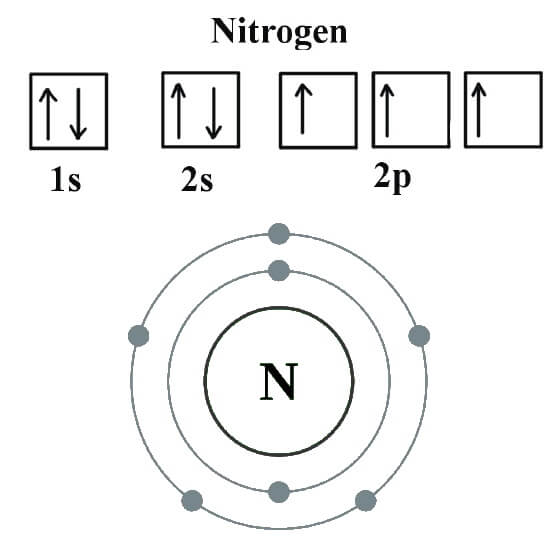
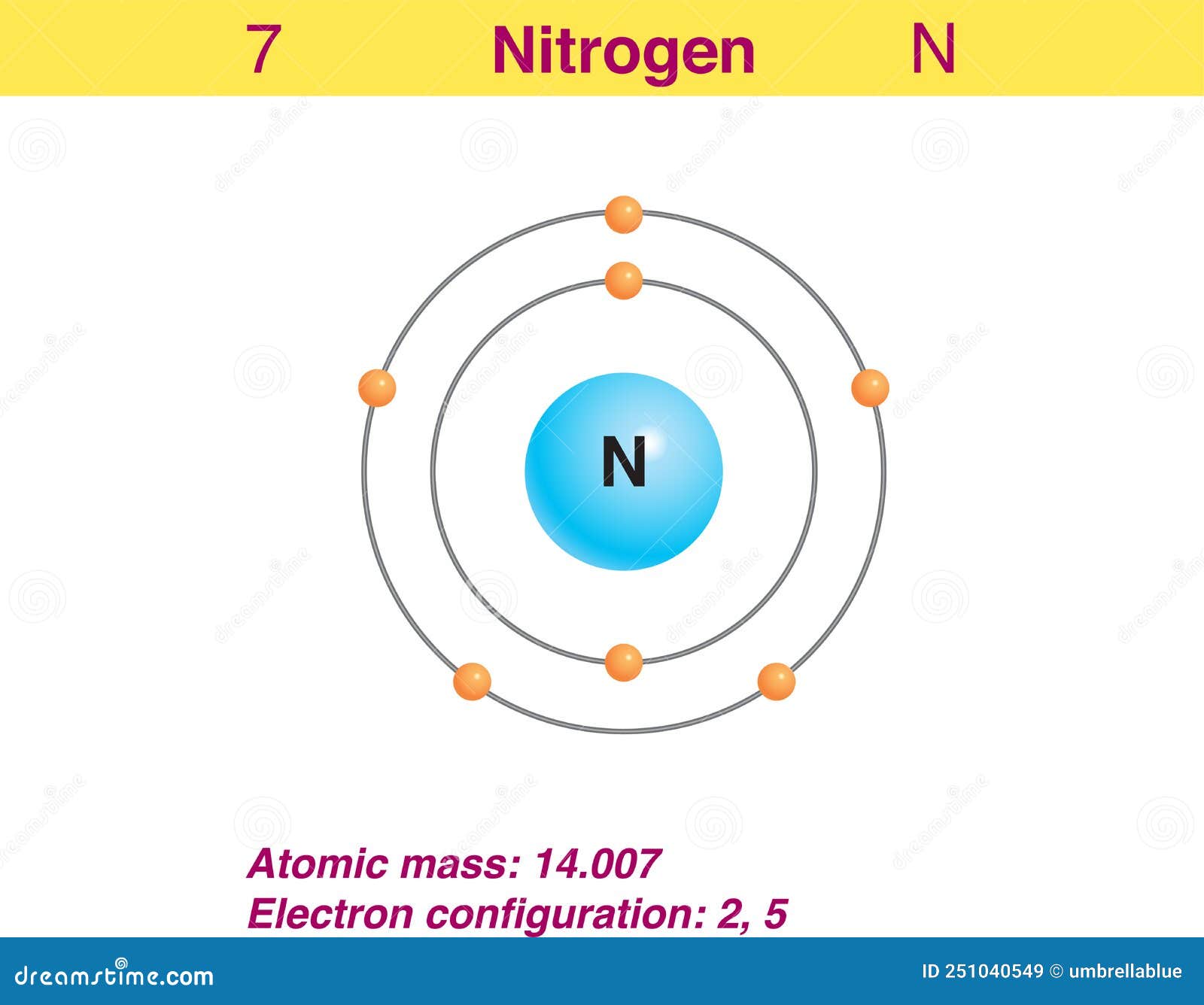
The electron configuration of nitrogen is 1s 2 2s 2 2p 3, helps to express how the electrons are arranged in nitrogen's atomic orbital.Let us study its electron configuration in detail. Nitrogen, a non-metal is found to be the lightest element of group 15 of periodic table.It is the 4 th highest electronegative element. It is a p-block element.
How many valence electrons does nitrogen have? Ask4Essay
1 to 20: Predicting an electron arrangement The electron arrangement of an atom can be predicted from its atomic number. For example, the atomic number of sodium is 11. Sodium atoms have 11.
Electron arrangements
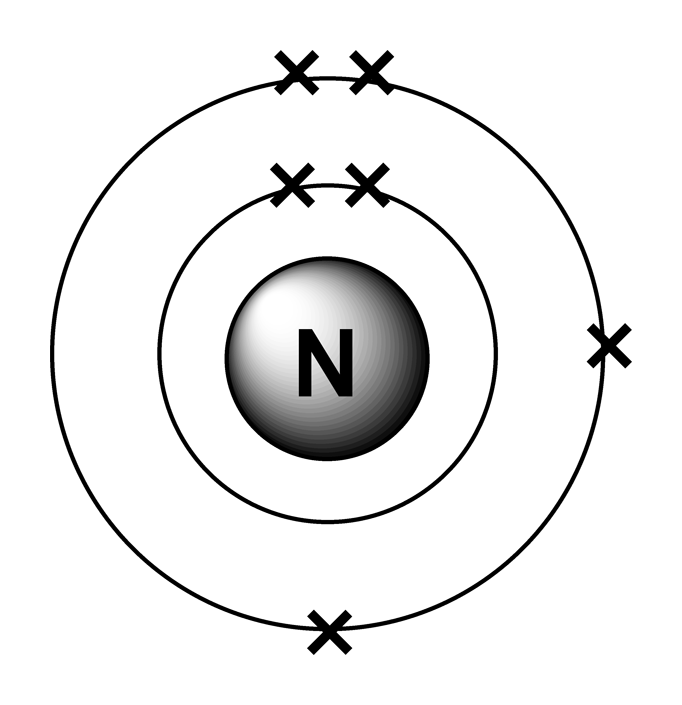
An electron configuration diagram is a model that depicts the position of electrons as they orbit the nucleus of an atom. Electrons are represented by dots or crosses and are positioned in energy levels, or 'shells', around the central nucleus. This is sometimes called the Bohr, or the 'solar system', model. Download this
Nitrogen Facts, Symbol, Discovery, Properties, Uses
The electron configuration of an atom is the representation of the arrangement of electrons distributed among the orbital shells and subshells. Commonly, the electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to represent an atom that has ionized into a cation or anion by compensating.
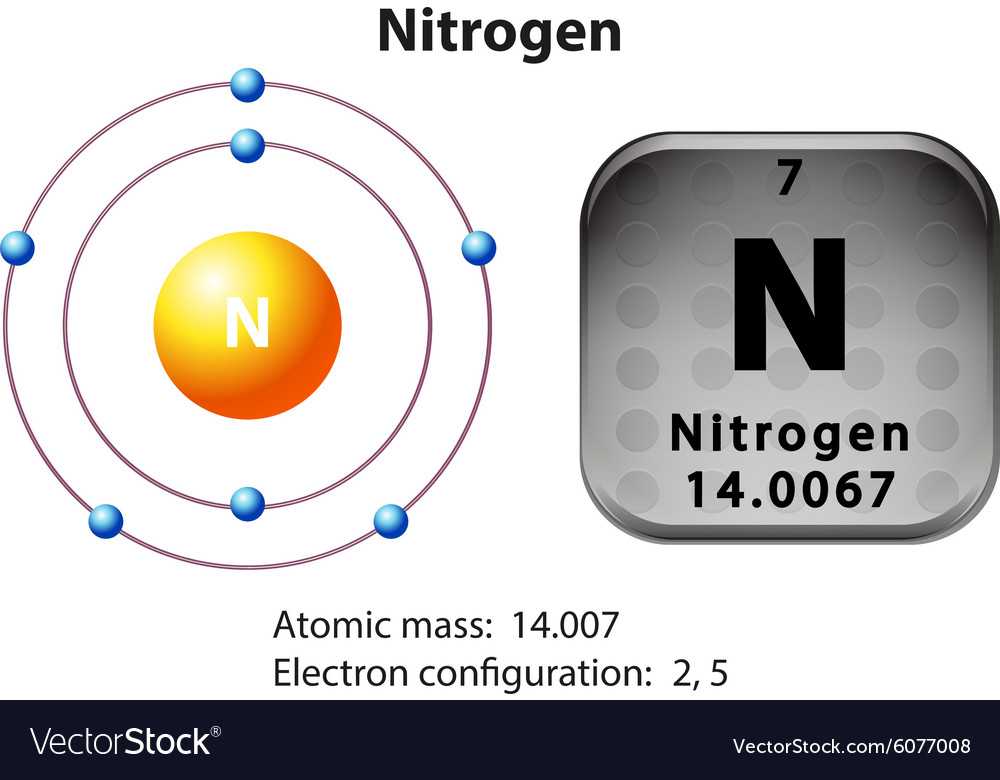
Symbol and electron diagram for nitrogen Vector Image
Campaigning & outreach Journals, books & databases News & events Locations & contacts Element Nitrogen (N), Group 15, Atomic Number 7, p-block, Mass 14.007. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images.
Nitrogen Element With Reaction, Properties, Uses, & Price Periodic Table
Nitrogen can also form ions by gaining or losing electrons. The Lewis dot structure of a nitrogen ion depends on the charge it carries. For example, the nitrogen ion with a positive charge (N+) has lost one electron and is represented by nitrogen's atomic symbol with a positive sign.On the other hand, the nitrogen ion with a negative charge (N-) has gained one electron, and its Lewis dot.
Electron of the Element Nitrogen Stock Vector Illustration of print
The electron configuration of an element is the arrangement of its electrons in its atomic orbitals. By knowing the electron configuration of an element, we can predict and explain a great deal of its chemistry.. The electron configuration of nitrogen is thus 1s 2 2s 2 2p 3. At oxygen, with Z = 8 and eight electrons, we have no choice. One.

Diagram representation of the element nitrogen Vector Image
In the quantum-mechanical model of an atom, electrons in the same atom that have the same principal quantum number (n) or principal energy level are said to occupy an electron shell of the atom. Orbitals define regions in space where you are likely to find electrons. As shown in Figure 3.7.2 3.7. 2 s orbitals are spherical shaped, and p.

How to find Valency? What are valence electrons? Teachoo
The arrangement of electrons in nitrogen in specific rules in different orbits and orbitals is called the electron configuration of nitrogen. The electron configuration of nitrogen is [ He] 2s 2 2p 3 , if the electron arrangement is through orbitals. Electron configuration can be done in two ways.

How to write the Electronic Configuration of Nitrogen Chemical
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 6.25 ): The number of the principal quantum shell, n,

Orbital Diagram For Nitrogen (N) Nitrogen Electron Configuration
Figure 4.8: Electron arrangement of a fluorine atom. Neon. Neon (\(\text{Ne}\)) has an atomic number of 10, meaning that a neutral atom also has 10 electrons. The first 2 electrons are found in the first energy level and the last 8 are found in the second energy level. . Figure 4.9: Electron arrangement of a neon atom.

What is the number of valence electrons in nitrogen? What's Insight
A Lewis Structure is a depiction of the arrangement of electrons in the standalone atoms of an element. In the Lewis Structure, electrons are depicted as dots.. Nitrogen's valence electron count, however, is 5, owing to its position in the 5th group of the periodic table. The plus sign denotes the absence of 1 electron; therefore, it is.

Electron Configuration (Elements 120) Good Science
Answers. Electrons are organized into shells and subshells around nuclei. The electron configuration states the arrangement of electrons in shells and subshells. Valence electrons are in the highest-numbered shell; all other electrons are core electrons.
Electron Configuration for Nitrogen (N and N3 ion)
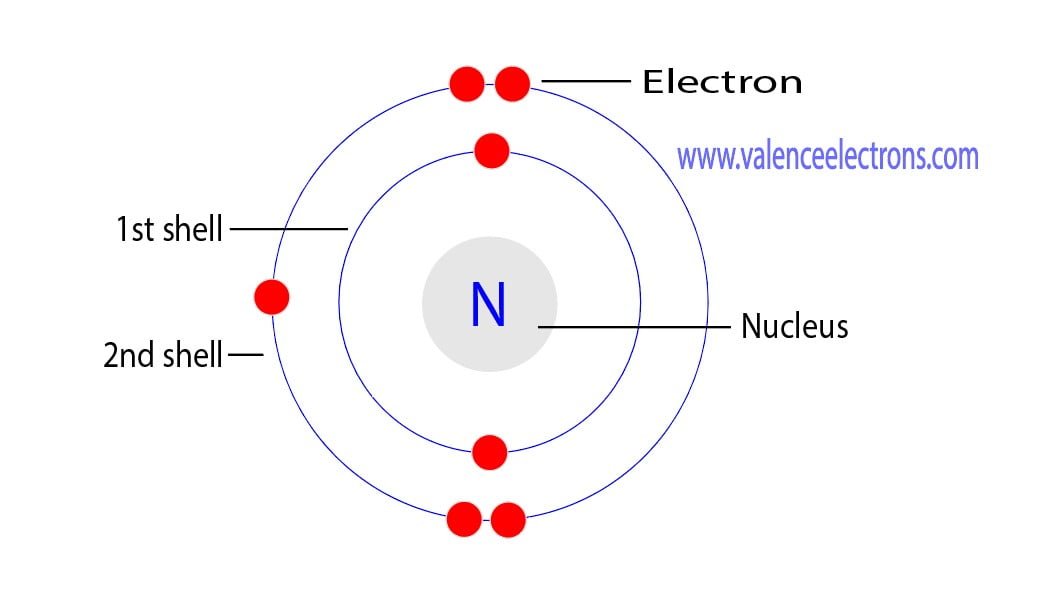
2.5: Arrangement of Electron (Shell Model) An electron shell is the outside part of an atom around the atomic nucleus. It is a group of atomic orbitals with the same value of the principal quantum number n n. Electron shells have one or more electron subshells, or sublevels. The name for electron shells comes from the Bohr model, in which.

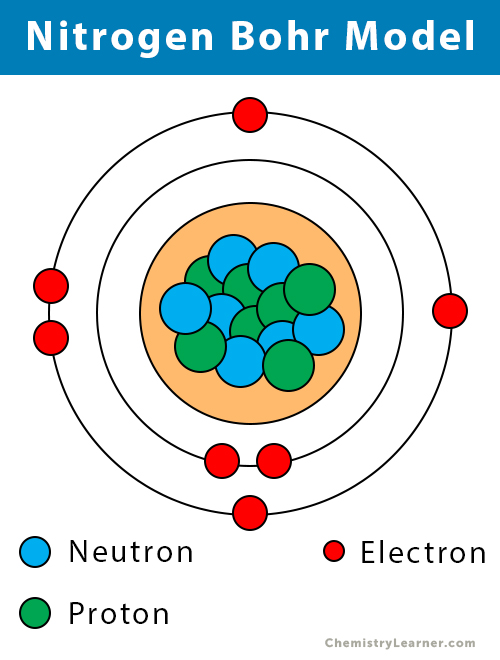
Bohr model Description & Development Britannica
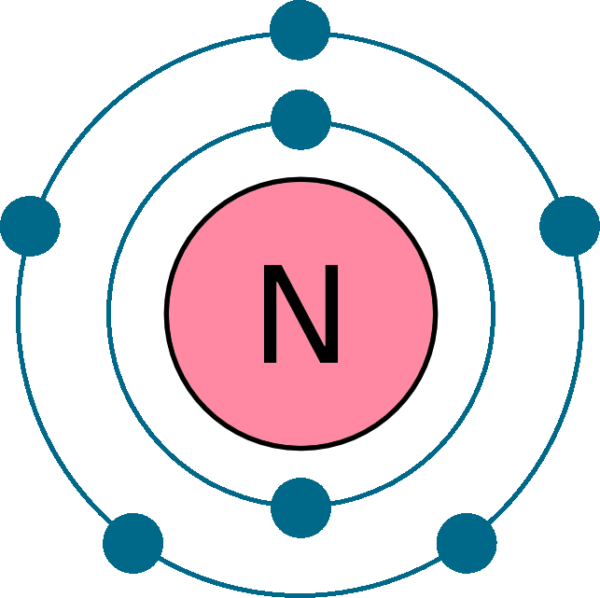
This electron arrangement indicates that the outermost orbit of Nitrogen (N) has 5 electrons. Hence, it lies in group 15. Why is Nitrogen in Period 2? Let me ask you a question. How many shells does Nitrogen have? It's 2. Right? You have already seen the bohr model of nitrogen element in the above table.

Nitrogen, atomic structure Stock Image C018/3688 Science Photo Library
The electrons in the outermost shell which determine the valency are known as valence electrons. Lewis Structure is a diagrammatic representation of any given molecule with the help of the constituent atoms and the position and arrangement of electrons to form bonds and lone pairs.