The Lewis structure of cyanide ion is shown below Wh… SolvedLib
Step 1: The foremost step of creating a Lewis structure is finding the valence electrons. Here we have to find the valence electrons of all three atoms, hydrogen, carbon, and nitrogen.
HCN Lewis StructureHydrogen Cyanide (HCN) Lewis Dot StructureDraw Lewis Structure of HCN
Chemistry learning made easy.This tutorial will help you deal with the lewis structure and moleculargeometry for hydrogen cyanide (HCN).
HCN Molecular Geometry YouTube
To start with making the Lewis Structure of HCN, we will first determine the central atom. And then place the remaining atoms in the structure. As Carbon is the least electronegative atom in this molecule, it will take the central position. Place the Hydrogen and Nitrogen atoms on both terminal sides of the Carbon like this:

HCN Lewis Structure, Molecular Geometry, Hybridization, MO Diagram, and Polarity Techiescientist
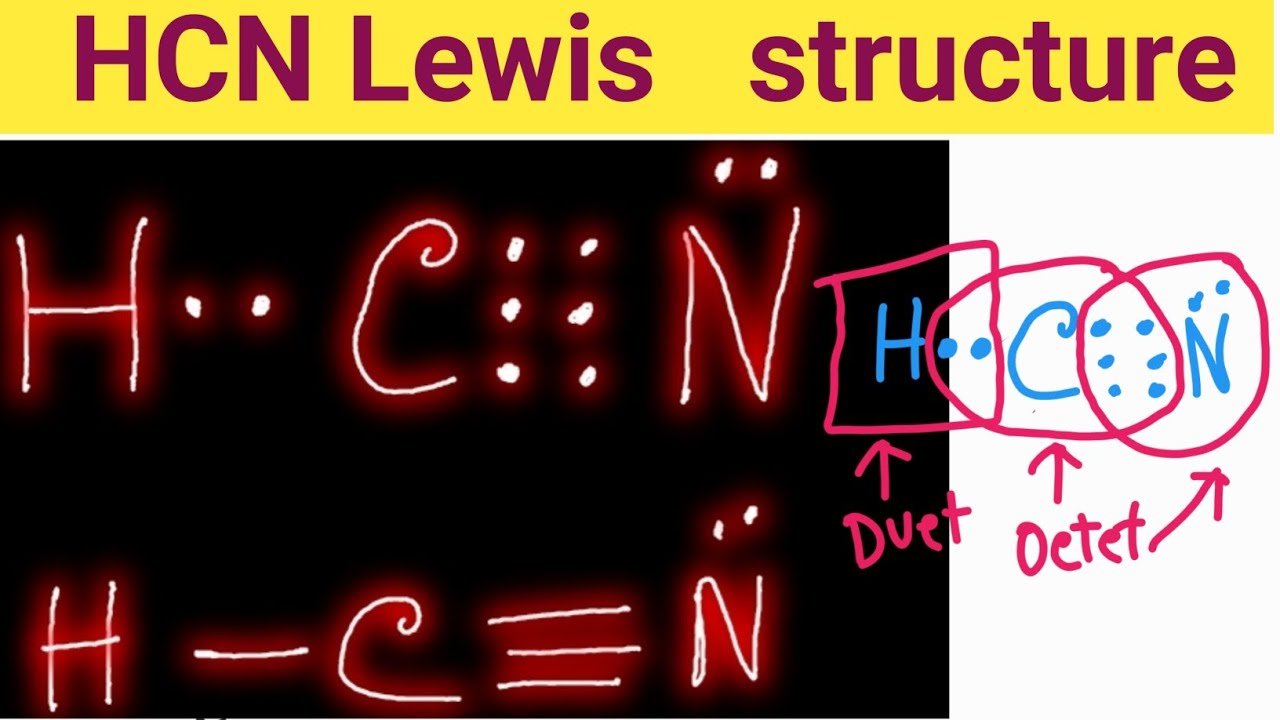
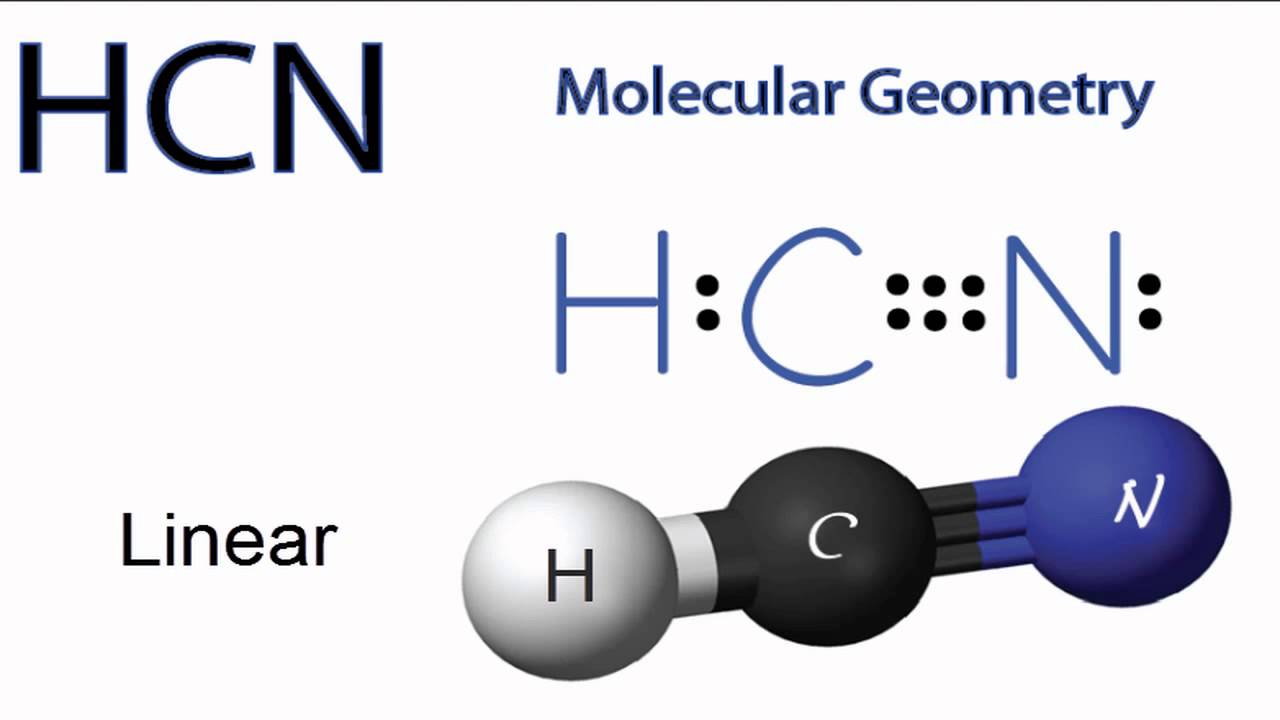
The Lewis structure or dot structure of a hydrogen cyanide (HCN) molecule can easily represent the electron arrangement around the atoms of HCN as it is made by bonding 3 different atoms together. HCN is a linear molecule which is bounded by a hydrogen atom, a carbon atom, and a nitrogen atom.
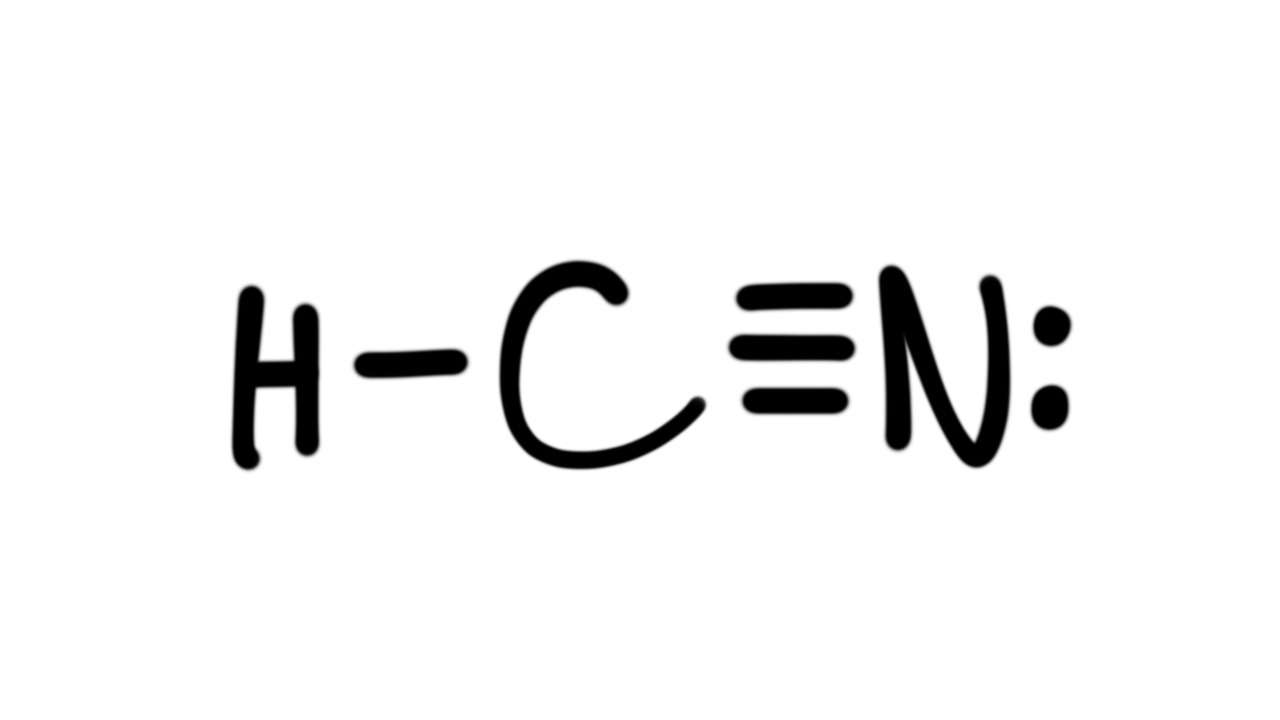
Hydrogen Cyanide YouTube
Lewis Structure of Hydrogen cyanide - HCN Chemical data of Hydrogen Cyanide-HCN Physical Properties of Hydrogen cyanide-HCN Chemical Properties of Hydrogen cyanide-HCN Hydrocyanic acid reacts with bases like sodium hydroxide forms sodium cyanide and water. The chemical equation is given below. HCN + NaOH → NaCN + H2O

Hydrogen Cyanide Lewis Structure
Lewis Structure and Hybridization of HCN (hydrocyanic acid, hydrogen cyanide) HCN has a hydrogen atom single-bonded to a carbon atom, and that carbon atom is triple-bonded to a nitrogen atom. These are all non-metals, so the bonds are covalent and HCN is therefore a covalent (aka Molecular) structure.

Chemical Structure Hydrogen Cyanide Hcn Stock Vector (Royalty Free) 2125623392 Shutterstock
Hydrogen cyanide is a one- carbon compound consisting of a methine group triple bonded to a nitrogen atom It has a role as a human metabolite, an Escherichia coli metabolite and a poison. It is a hydracid and a one- carbon compound. It is a conjugate acid of a cyanide. It is a tautomer of a hydrogen isocyanide. ChEBI

Lewis structure of HCN (Hydrogen cyanide) YouTube
The Lewis Structure of HCN. When creating the Lewis structure of HCN, it is important to start by identifying the central atom, which in this case is carbon. Nitrogen and hydrogen atoms are then placed around the carbon atom, with the hydrogen atoms forming a single bond with the carbon atom and the nitrogen atom forming a triple bond with the.

Draw the Lewis dot structure of Hydrogen cyanide (HCN) molecule
A triple bond forms when three electron pairs are shared by a pair of atoms, as in carbon monoxide (CO) and the cyanide ion (CN -): Writing Lewis Structures with the Octet Rule. For very simple molecules and molecular ions, we can write the Lewis structures by merely pairing up the unpaired electrons on the constituent atoms. See these examples:
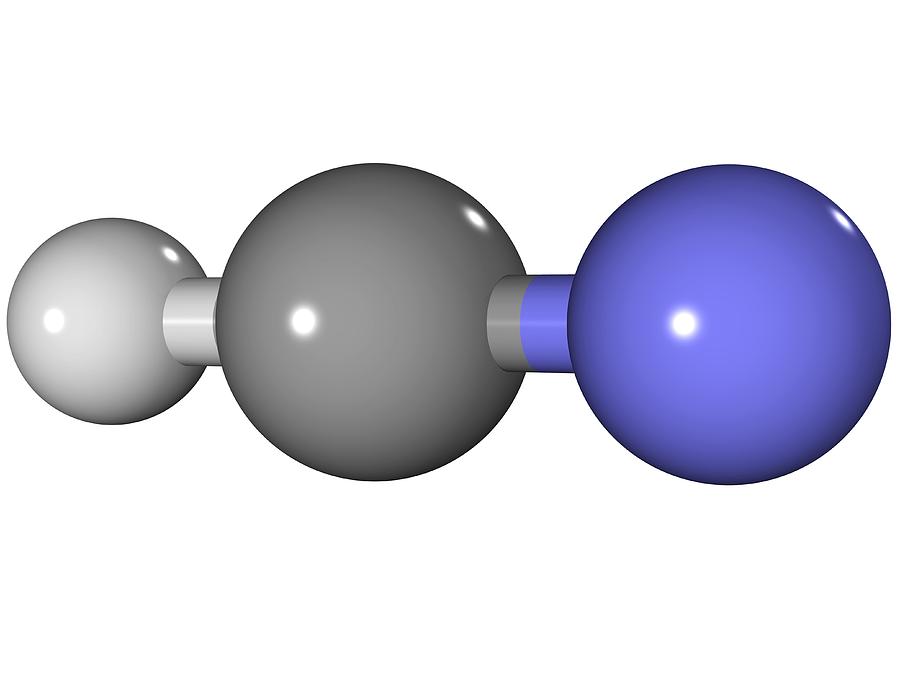
Hydrogen Cyanide Molecule Photograph by Laguna Design Pixels
Lewis Structure of HCN chemistNATE 260K subscribers Subscribe Subscribed 457K views 10 years ago Lewis Structures The Lewis Structure (Lewis Dot Diagram) for HCN. 1. Count electrons 2..

Bond formation, hydrogen cyanide molecule Stock Image C028/6480 Science Photo Library
Hey Guys!In this video, we will look at the Lewis Structure of Hydrogen Cyanide having a chemical formula of HCN. The molecule is made up of one Hydrogen ato.
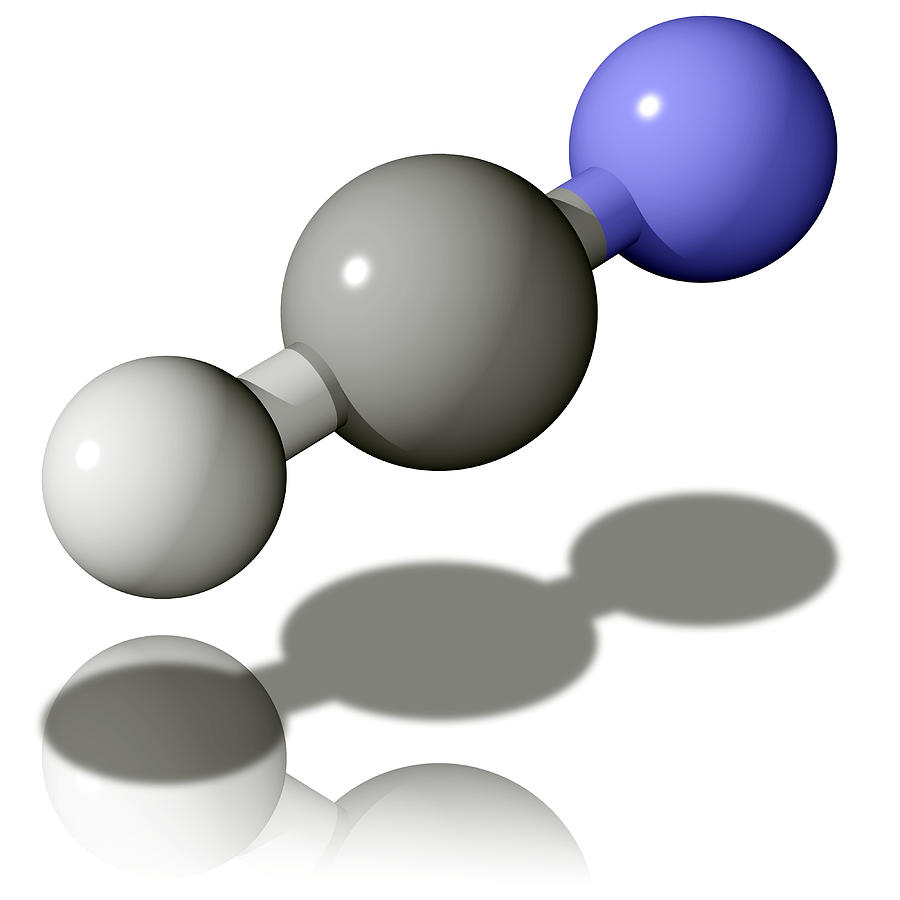
Hydrogen Cyanide Molecule Photograph by Laguna Design Pixels
5.3C: HCN H C N. Page ID. HCN, hydrogen cyanide, is a volatile and poisnous compound with distinguished bitter odor. It is linear molecule with a triple bond between C and N atom and has bond angle of 180 degrees. It can be found in fruits that have pits due to the fact that they contain small amounts of cyanohydrins which slowly releases.

Hcn hydrogen cyanide molecule Royalty Free Vector Image
Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure.".
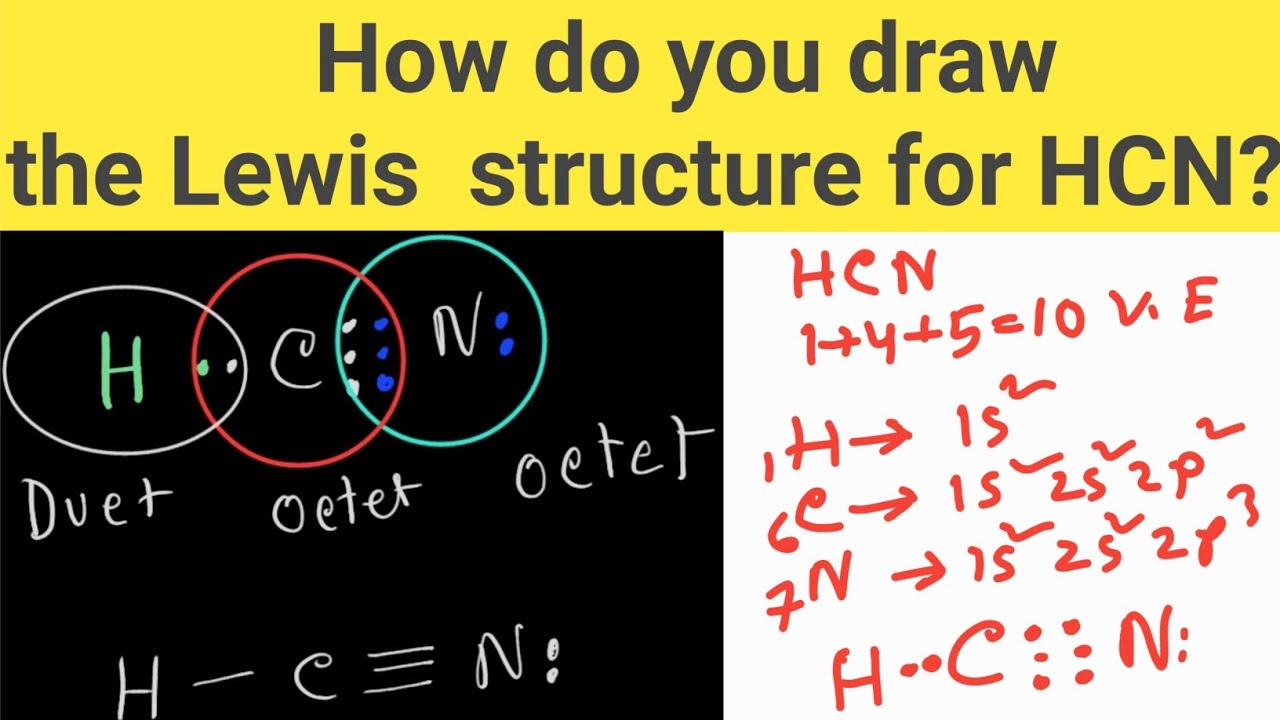
How do you draw the Lewis structure of HCN (hydrogen cyanide)? HCN Lewis Dot Structure YouTube
2. Each hydrogen atom (group 1) has one valence electron, carbon (group 14) has 4 valence electrons, and oxygen (group 16) has 6 valence electrons, for a total of [ (2) (1) + 4 + 6] = 12 valence electrons. 3. Placing a bonding pair of electrons between each pair of bonded atoms gives the following: Six electrons are used, and 6 are left over.
[Solved] Draw the Lewis structure for it as well. 3. Hydrogen cyanide (HCN)... Course Hero
We can draw Lewis structures for polyatomic ions (ions containing multiple atoms) using the same stepwise procedure as for neutral molecules. In this video, we'll see how to construct the Lewis diagram of the cyanide ion (CN⁻). Created by Sal Khan. Questions Tips & Thanks Want to join the conversation? Sort by: Top Voted Maria Gregory 3 years ago
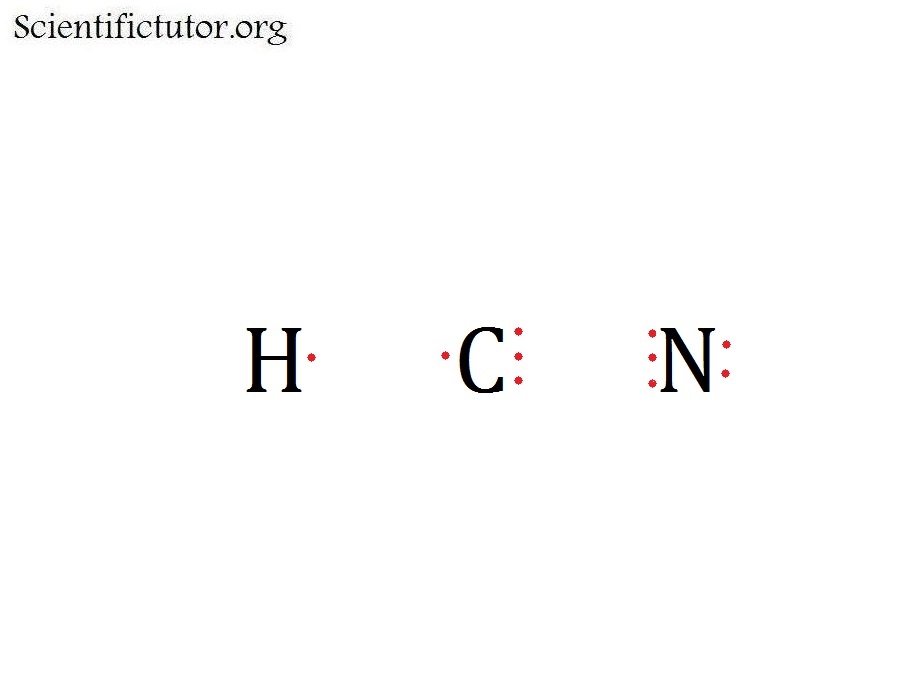
Hcn Dot Diagram
There is a single covalent bond between the hydrogen and carbon atom, represented by two dots, :, each of which represents a shared electron; a triple covalent bond between the carbon and nitrogen atom, represented by three pairs of dots, :::, representing three pairs of shared electrons, and a lone pair of electrons on the nitrogen atom, repres.