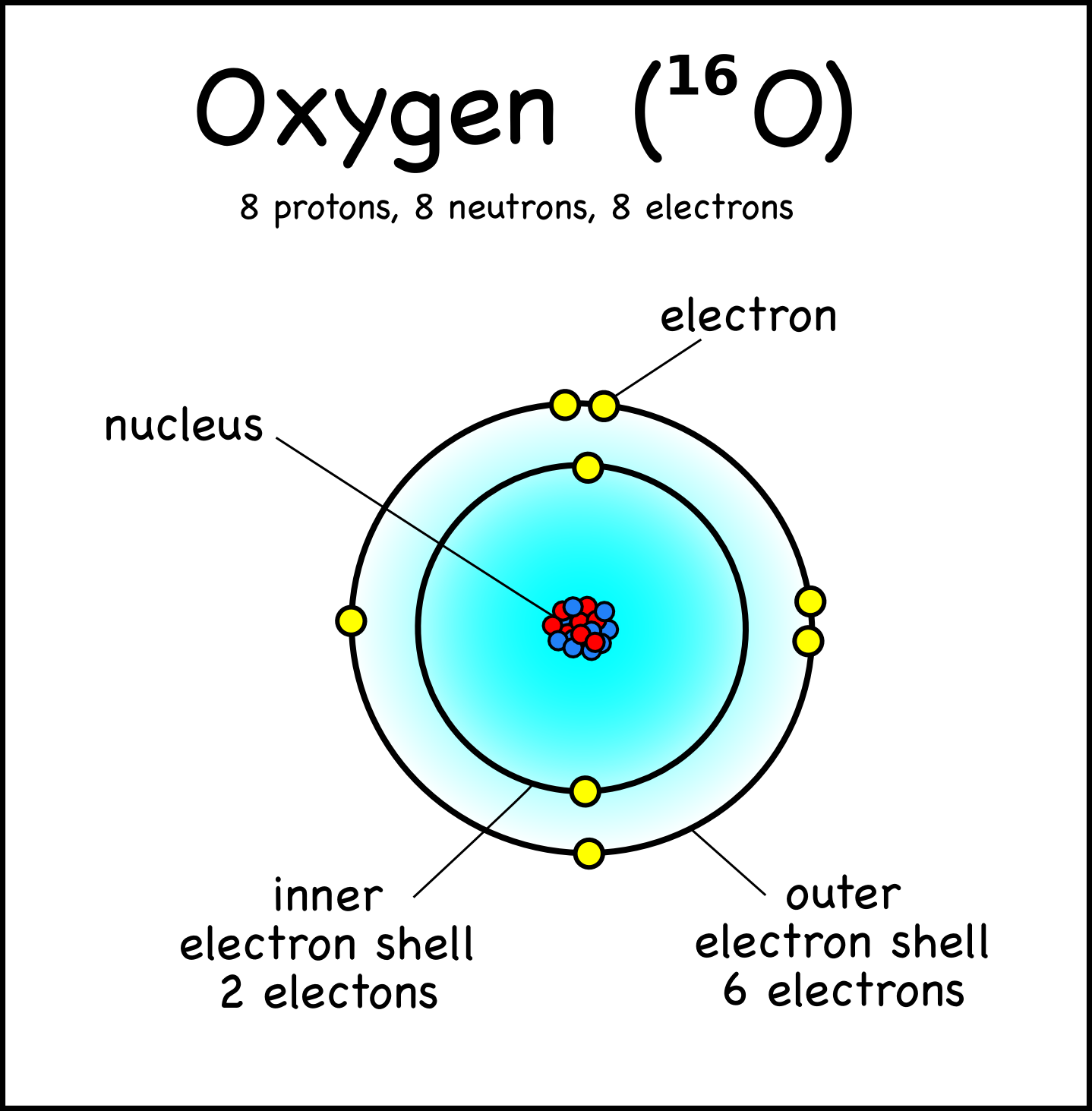
Drawing Atoms Montessori Muddle
Explanation: the configuration of o is 2s22p4. so by gaining 2 electrons it is stabilised by completing octet. Answer link. the electronic configuration of O^2- is 2s ^2 2p^6. the configuration of o is 2s^2 2p^4. so by gaining 2 electrons it is stabilised by completing octet.
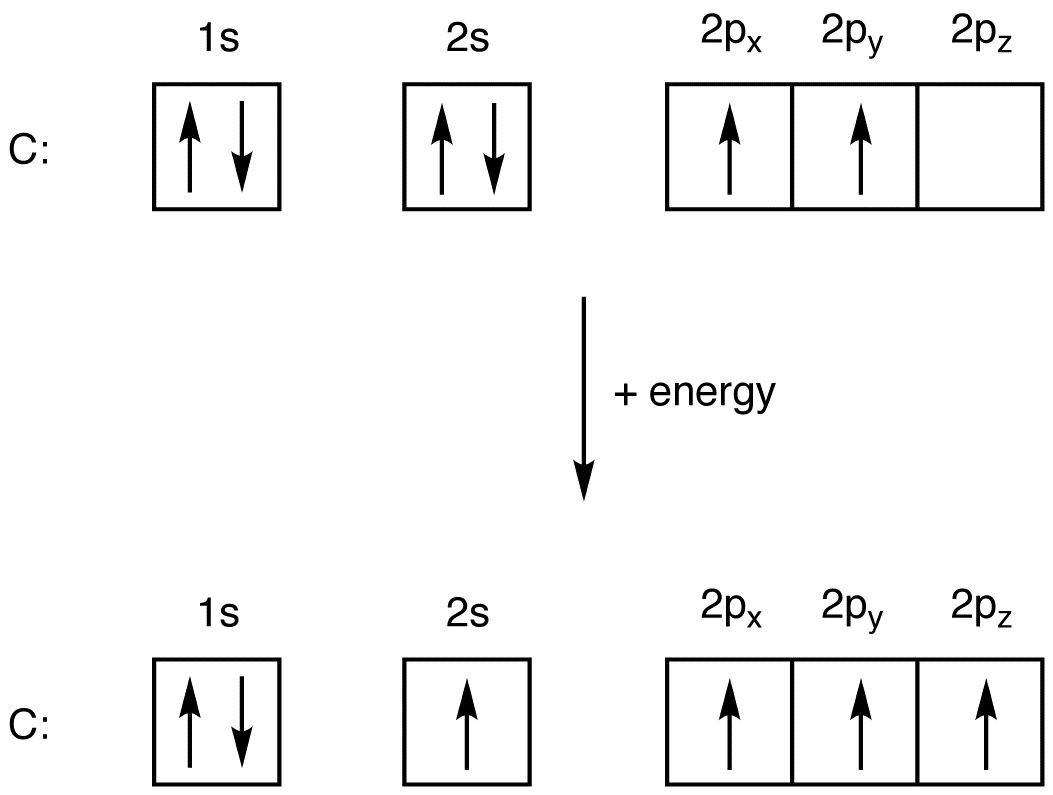
Oxygen Electron Configuration (O) with Orbital Diagram
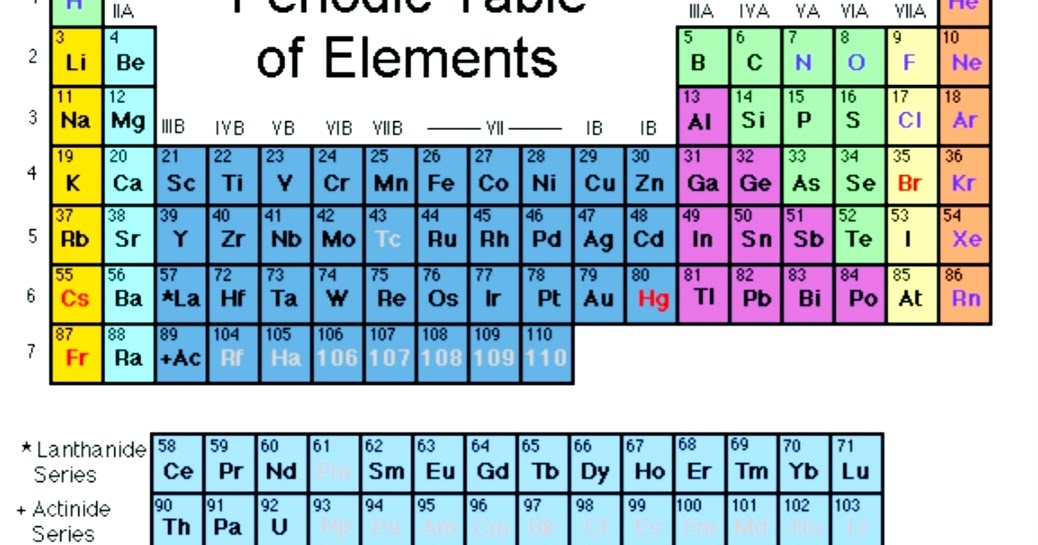
Oxygen is an element having an atomic number 8 and an atomic symbol O. It belongs to Group-16 and second period. It is a highly reactive non-metal. Electronic configuration: The arrangement of electrons into the orbitals of an atom using some fundamental principle is called its electronic configuration. Electronic configuration of Oxygen:
Electron Configuration for Oxygen (O, and O2 ion)
Electron configurations are the summary of where the electrons are around a nucleus. As we learned earlier, each neutral atom has a number of electrons equal to its number of protons.

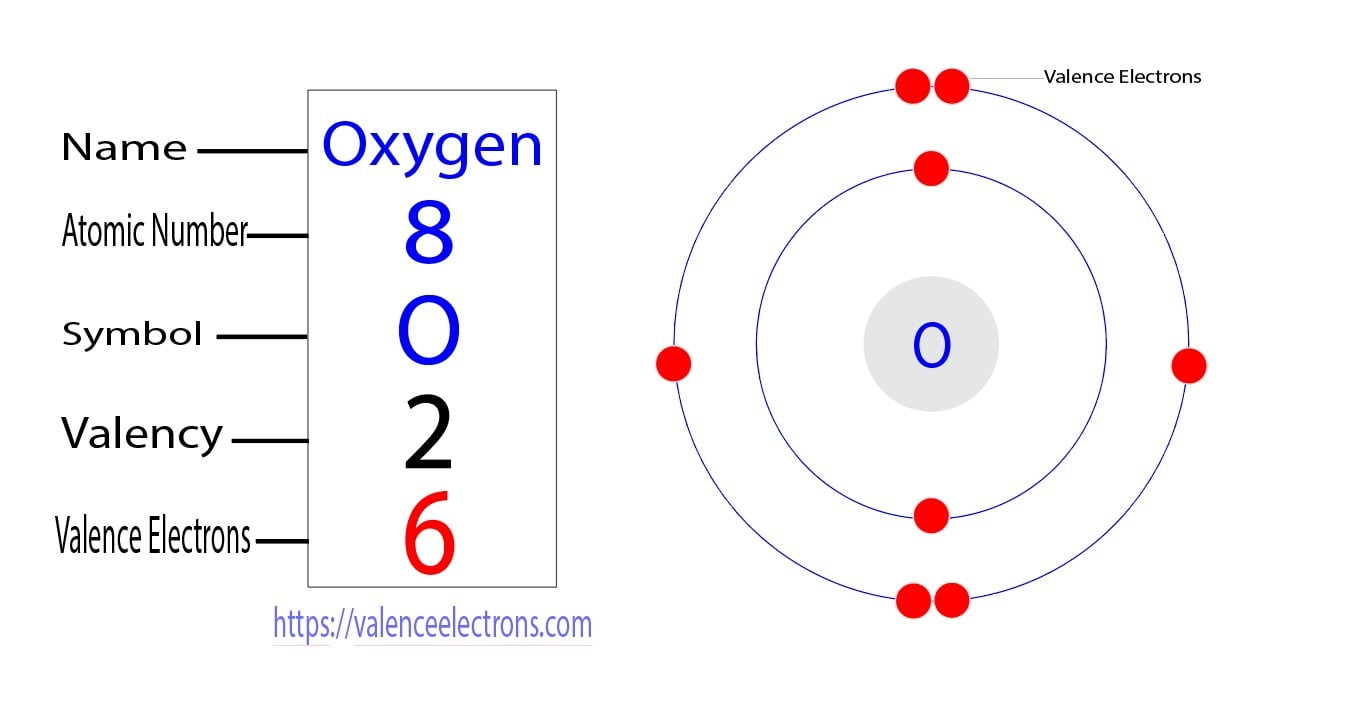
Symbol and electron diagram for Oxygen Royalty Free Vector
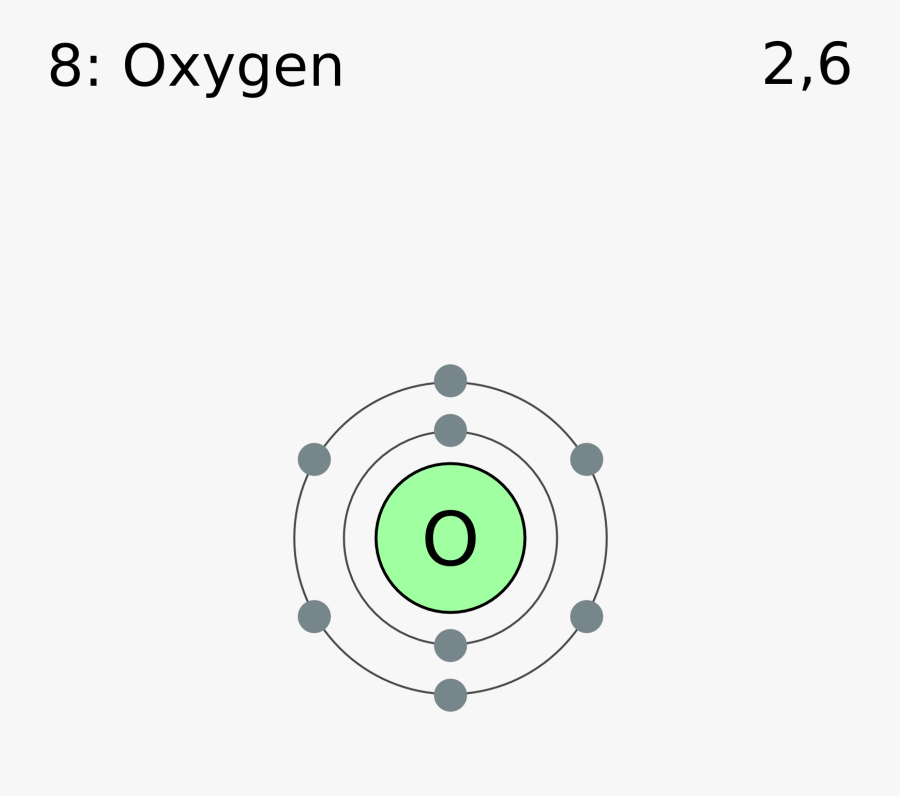
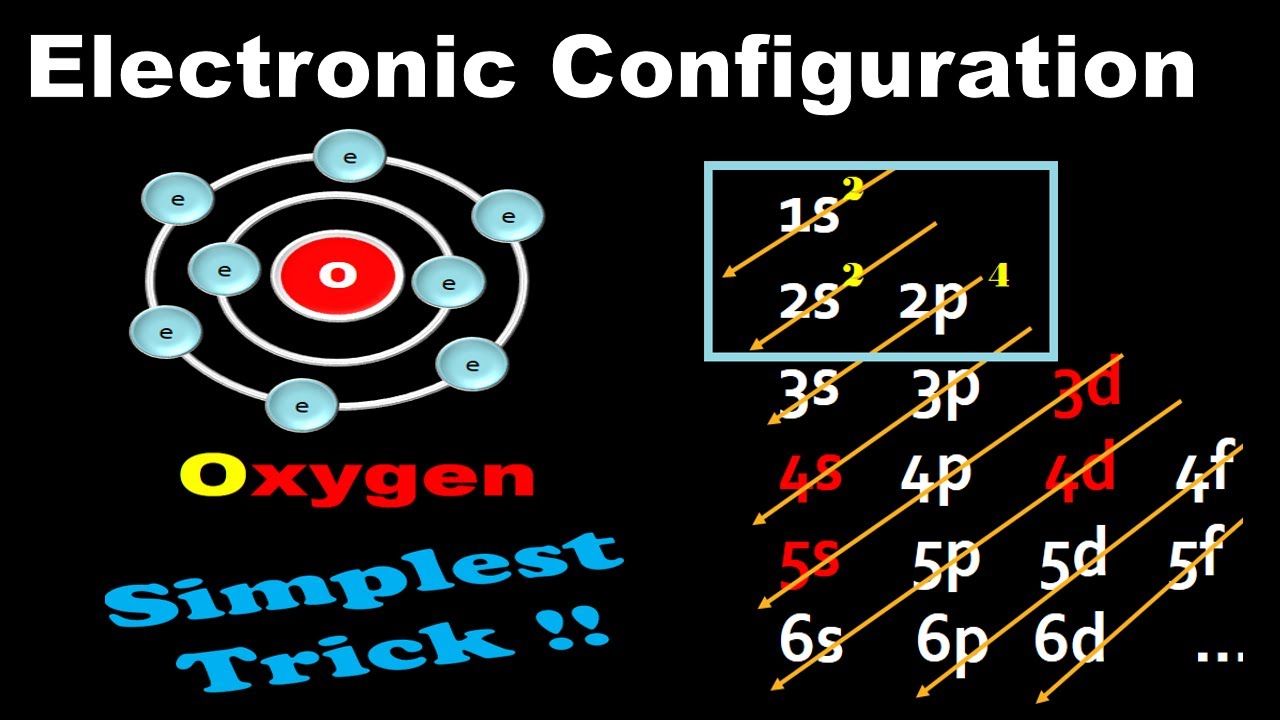
Electron Configuration Notation: -shows the arrangment of electrons around the nucleus of an atom. - helps chemist understanding how elements form chemical bonds. - can be written using the period table or an electron configuration chart. How to Write the Electron Configuration for Oxygen Oxygen is the eighth element with a total of 8 electrons.

Electron Configuration Of Oxygen In Ground State
The O atom has 2s 2 2p 4 as the electron configuration. Therefore, O has 2 unpaired electrons. Answer (b): The Br atom has 4s 2 3d 10 4p 5 as the electron configuration. Therefore, Br has 1 unpaired electron. Answer (c): The B atom has 2s 2 2p 1 as the electron configuration. Because it has one unpaired electron, it is paramagnetic.
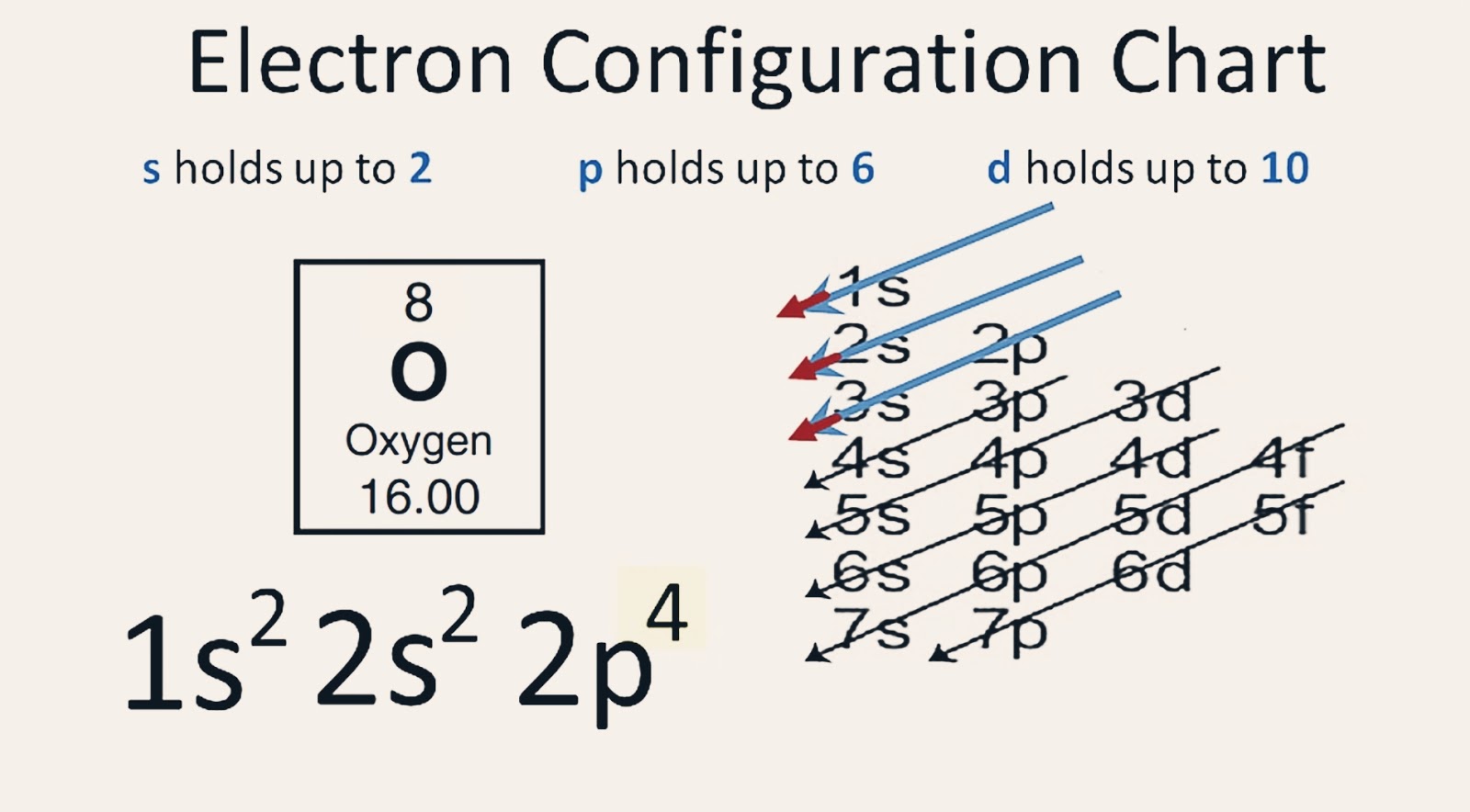
【5 Steps】Oxygen Electron Configuration in Just 5 Steps Electron Configuration of Oxygen(O)
Electron Configurations. The electron configuration of an atom is the representation of the arrangement of electrons distributed among the orbital shells and subshells. Commonly, the electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to represent an atom that has ionized into a cation or anion by compensating with the loss of or gain.
【5 Steps】Oxygen Electron Configuration in Just 5 Steps Electron Configuration of Oxygen(O)
In this case, 2+2+6+2+6+2+10+6+2+1= 39 and Z=39, so the answer is correct. A slightly more complicated example is the electron configuration of bismuth (symbolized Bi, with Z = 83). The periodic table gives the following electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p65s2 4d10 5p6 6s2 4f14 5d10 6p3.
Electron Shell Diagram For Oxygen , Free Transparent Clipart ClipartKey
The electronic configuration of an atom in the shell atomic model may be expressed by indicating the number of electrons in each shell beginning with the first. For example, sodium (atomic number 11) has its 11 electrons distributed in the first three shells as follows: the K and L shells are completely filled, with 2 and 8 electrons.
Electronic Configuration For Oxygen spdf Trick Chemistry Atomic Number 8 YouTube
1). You can effortlessly find every single detail about the elements from this single Interactive Periodic table. 2). You will get the detailed information about the periodic table which will convert a newbie into pro. 3). You will also get the HD images of the Periodic table (for FREE).
How to Write Ground State Electron Configuration in Chemistry
799 130K views 4 years ago In this video we will write the electron configuration for O 2-, the Oxide ion. We'll also look at why Oxygen forms a 2- ion and how the electron configuration for.

The electron configuration of oxygen is 1s2,2s2 2p4. Science chemistry, Electron configuration
This electron configuration calculator will instantly show you the distribution of electrons in the orbitals of any periodic element you choose. Typically, you need at least 8 steps to determine the electron configuration, starting with finding the atomic number by looking at the list of orbitals and understanding the notation.
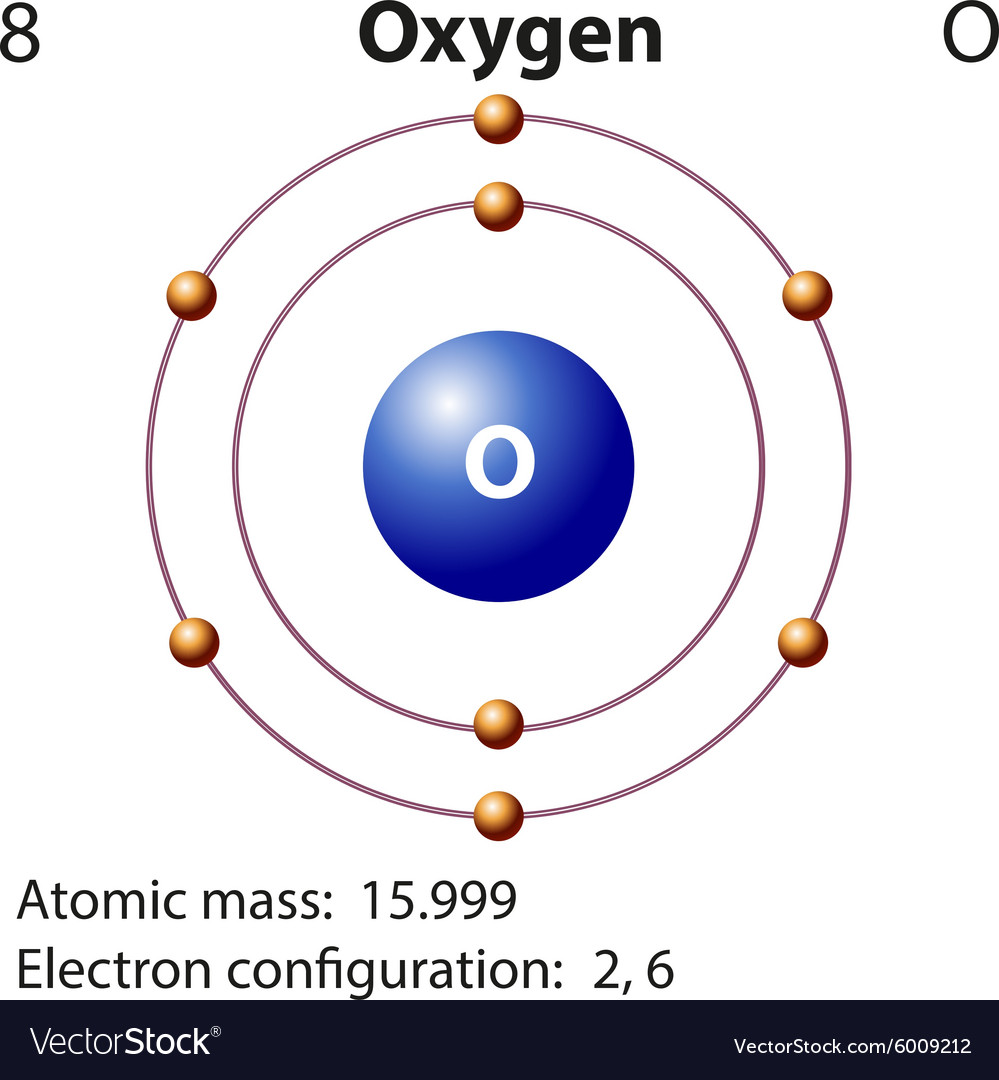
Diagram representation element oxygen Royalty Free Vector
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 6.25 ): The number of the principal quantum shell, n,

Electron configuration of oxygen ion Lousiana
Electron configurations are a simple way of writing down the locations of all of the electrons in an atom. As we know, the positively-charged protons in the nucleus of an atom tend to attract negatively-charged electrons.

Oxygen Bohr Model (Diagram, Steps To Draw) Techiescientist
AboutTranscript. Electron configurations describe where electrons are located around the nucleus of an atom. For example, the electron configuration of lithium, 1s²2s¹, tells us that lithium has two electrons in the 1s subshell and one electron in the 2s subshell. Created by Sal Khan.

List of Electron Configurations of Elements
Oxygen Electron Configuration Wayne Breslyn 724K subscribers Join Subscribe Subscribed 683 Share 130K views 10 years ago A step-by-step description of how to write the electron configuration.

Oxygen electronic configuration,how to Write Oxygen electronic configuration YouTube
The first two electrons in lithium fill the 1 s orbital and have the same sets of four quantum numbers as the two electrons in helium. The remaining electron must occupy the orbital of next lowest energy, the 2 s orbital (Figure 8.3. 3 or 8.3. 4 ). Thus, the electron configuration and orbital diagram of lithium are: